A pump is a device which increases the pressure of the working fluid. Pumps are are restricted to liquids because of inefficiencies and possibility of cavitation if the working fluid were to be a liquid-vapour mixture.
In the Rankine cycle, the pump isentropically compresses the liquid leaving the condenser at state 1 to the required pressure for the boiler at state 2.
Assuming an isentropic pump, adiabatic conditions, and steady state steady flow conditions, the first law energy balance gives:
![]()
![]()
![]()
Where wp is the work done by the pump. Note however, it is difficult to calculate h2 using conventional tables and methods since state 2 is in the sub-cooled region.
In most steam generating plants, the change in the specific volume of the liquid water from state 1 to state 2 is usually less than 1 percent. As a result, the working fluid can be considered incompressible. Assuming an isentropic pump, adiabatic conditions, and steady state steady flow conditions:
First Law:
![]()
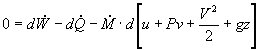
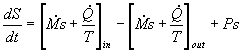
![]()
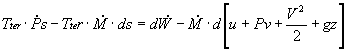
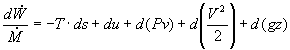
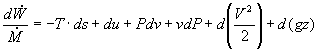
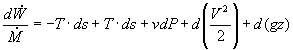
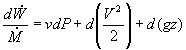
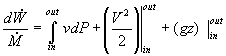
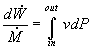
![]()
![]()
The isentropic efficiency of an adiabatic pump can be calculated as follows.
![]()
![]()
where
Ws = isentropic work by the turbine per unit mass flow
W = actual work by the turbine per unit mass flow
The isentropic work of the pump will always be less than the actual work, therefore the efficiency will always be less than 1.
It should be noted that the work required to compress a fluid is proportionally related to the specific volume. Therefore it would take considerably less work to compress a liquid than to compress a gas for the same change in pressure.
The boiler is a device which adds thermal energy to the working fluid. In a typical steam power plant, the heat added to the system may come from a variety of energy sources, such as nuclear, gas, and oil reactors, or even solar cells.
In the Rankine cycle, the boiler adds thermal energy to the work fluid from state 2 to state 3 at a constant pressure. The heat addition to the system can be obtained using the 1st law energy balance and assuming no work, steady state steady flow conditions, and negligible changes in kinetic and potential energy.
![]()
![]()
![]()
To optimize the efficiency of the Rankine cycle, the temperature of the boiler is usually set to the maximum temperature allowable by the material of the boiler.
Turbines are devices which convert steam into rotating energy by means of a set of blades connected to a shaft. Steam enters the turbine (state 3) at high pressure and expands. The expansion causes the rotor shaft to turn producing shaft work. The temperature of the working fluid leaving the turbine at state 4 drops significantly during this process.
The steam entering the turbine must be either dry saturated vapor or gas. High moisture content in the working fluid will cause cavitation.
In the Rankine cycle, the turbine is assumed to be isentropic, adiabatic, have negligible kinetic and potential energy, and have steady state steady flow conditions. The work produced by a turbine can be obtained using the 1st law analysis.
![]()
![]()
![]()
The isentropic efficiency of an adiabatic turbine can be calculated as follows.
![]()
![]()
where
Ws = isentropic work by the turbine per unit mass flow.
W = actual work by the turbine per unit mass flow.
The condenser is a device used to cool the working fluid by transferring heat to the environment. In typical steam generating plants, heat is transferred to a nearby lake or river.
In the Rankine cycle, the condenser isobarically changes the phase of the working fluid from a wet-vapour (state 4) to a saturated liquid (state 1). The heat transferred to the environment can be obtained by applying the 1st law of thermodynamics.
![]()
![]()
![]()
Based on the theory of the Carnot cycle, the efficiency of the Rankine cycle can be increased by either decreasing the temperature at which heat is rejected, or by increasing the temperature at which heat is added. Various processes that increase efficiency include reheating, regeneration, and superheat.
For first law efficiency:
![]()
![]()
![]()